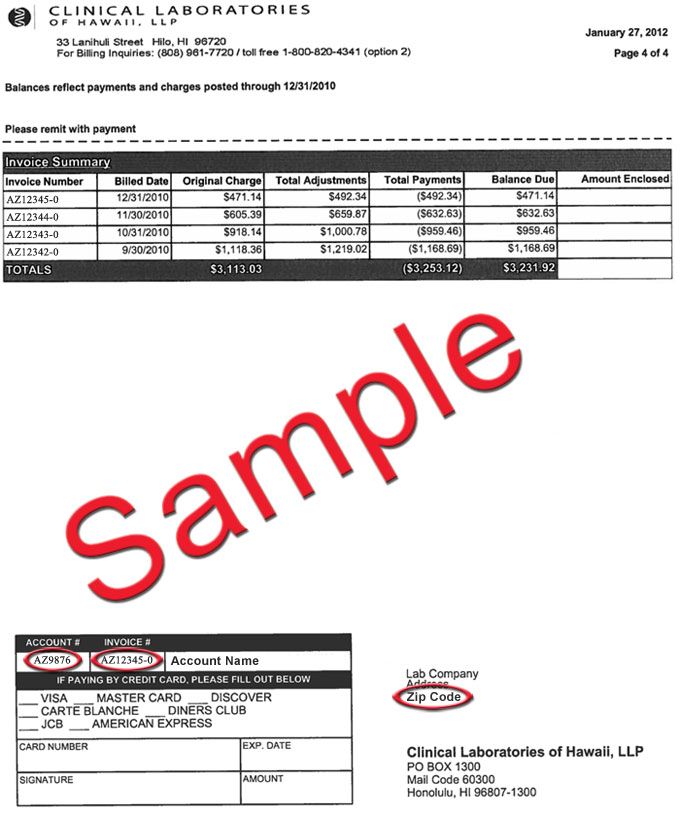
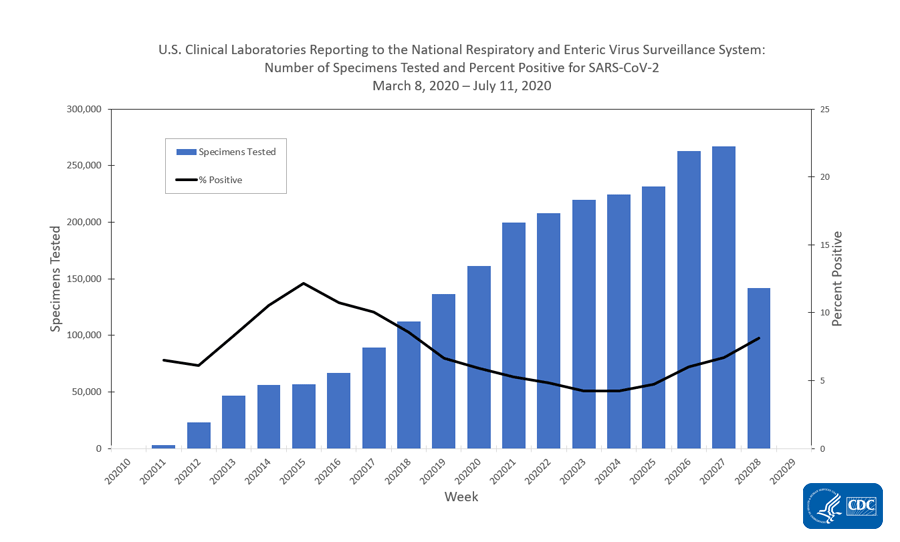
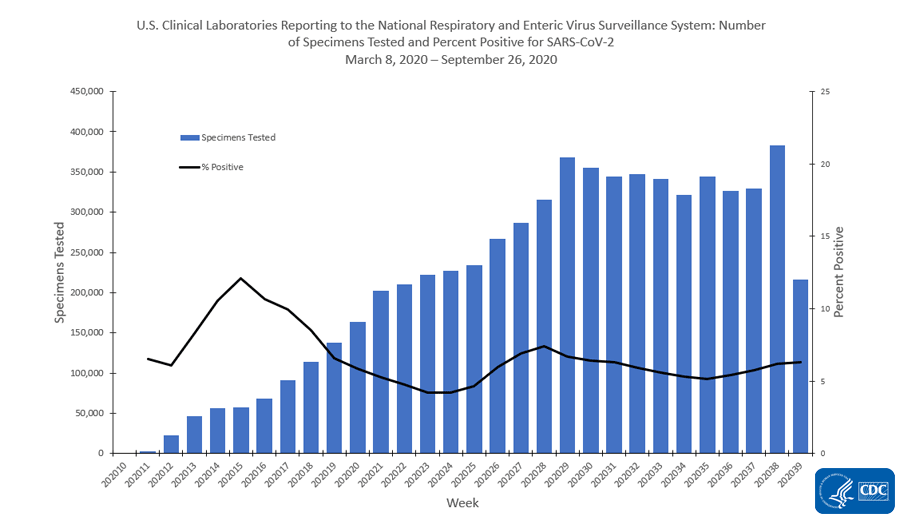
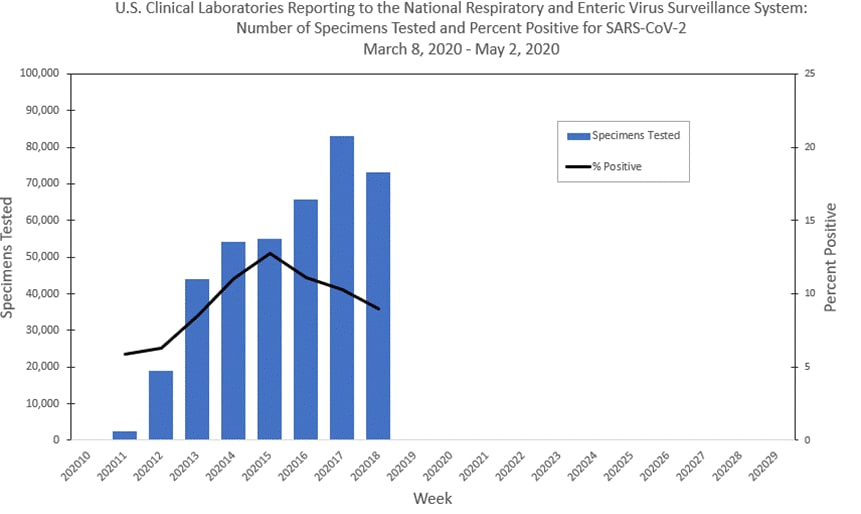
DATE: April 30, 2020 TO: Clinical Laboratories, Limited Service Labs, and Other Entities Supporting SAR- CoV-2 Testing FROM: Ne
Jurisdiction J Part B - Postpayment Review Results for Clinical Labs Definitive Drug Testing for 22 or More Drug Classes for October Through December 2022

TVH Laboratory Achieves Outstanding Results in Clinical Laboratory Improvement Amendments (CLIA) Inspection - Teton Valley Health
Eligible Hospital and Critical Access Hospital Meaningful Use Core Measures, Measure 8 of 16, Stage 2 (November, 2014)