
Hairy cell leukemia 2022: Update on diagnosis, risk‐stratification, and treatment - Troussard - 2022 - American Journal of Hematology - Wiley Online Library

Moxetumomab Pasudotox Pivotal Data in Patients with Previously-Treated Hairy Cell Leukemia Presented at the 2018 ASCO Meeting | Business Wire

Moxetumomab pasudotox in heavily pre-treated patients with relapsed/refractory hairy cell leukemia (HCL): long-term follow-up from the pivotal trial | Journal of Hematology & Oncology | Full Text

Deamidation in Moxetumomab Pasudotox Leading to Conformational Change and Immunotoxin Activity Loss - Journal of Pharmaceutical Sciences
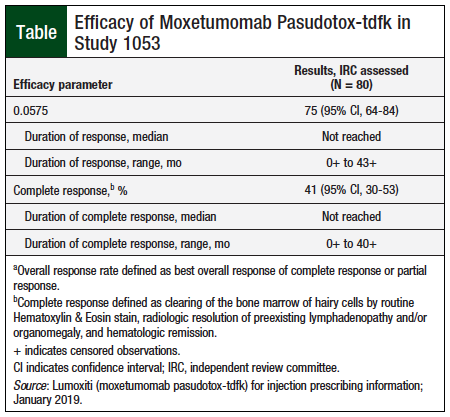
Lumoxiti (Moxetumomab Pasudotox-tdfk) First CD22-Directed Cytotoxin FDA Approved for Relapsed or Refractory Hairy-Cell Leukemia

References in Pharmacokinetic and Pharmacodynamic Comparability Study of Moxetumomab Pasudotox, an Immunotoxin Targeting CD22, in Cynomolgus Monkeys - Journal of Pharmaceutical Sciences
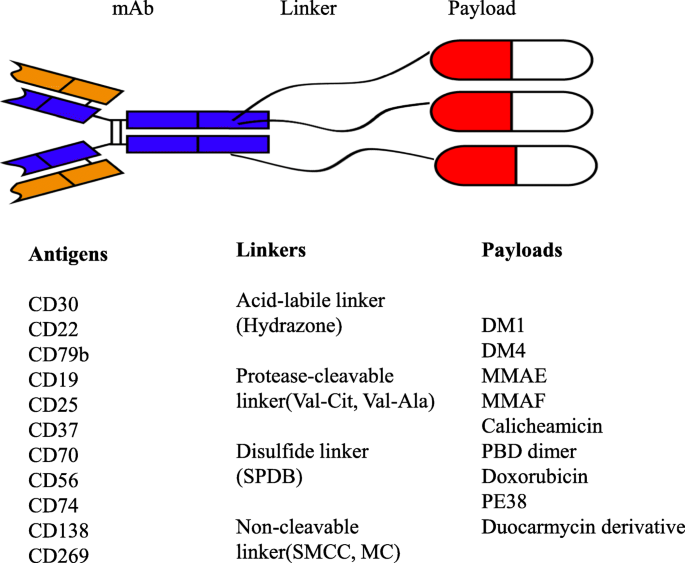
Antibody-drug conjugates in clinical trials for lymphoid malignancies and multiple myeloma | Journal of Hematology & Oncology | Full Text
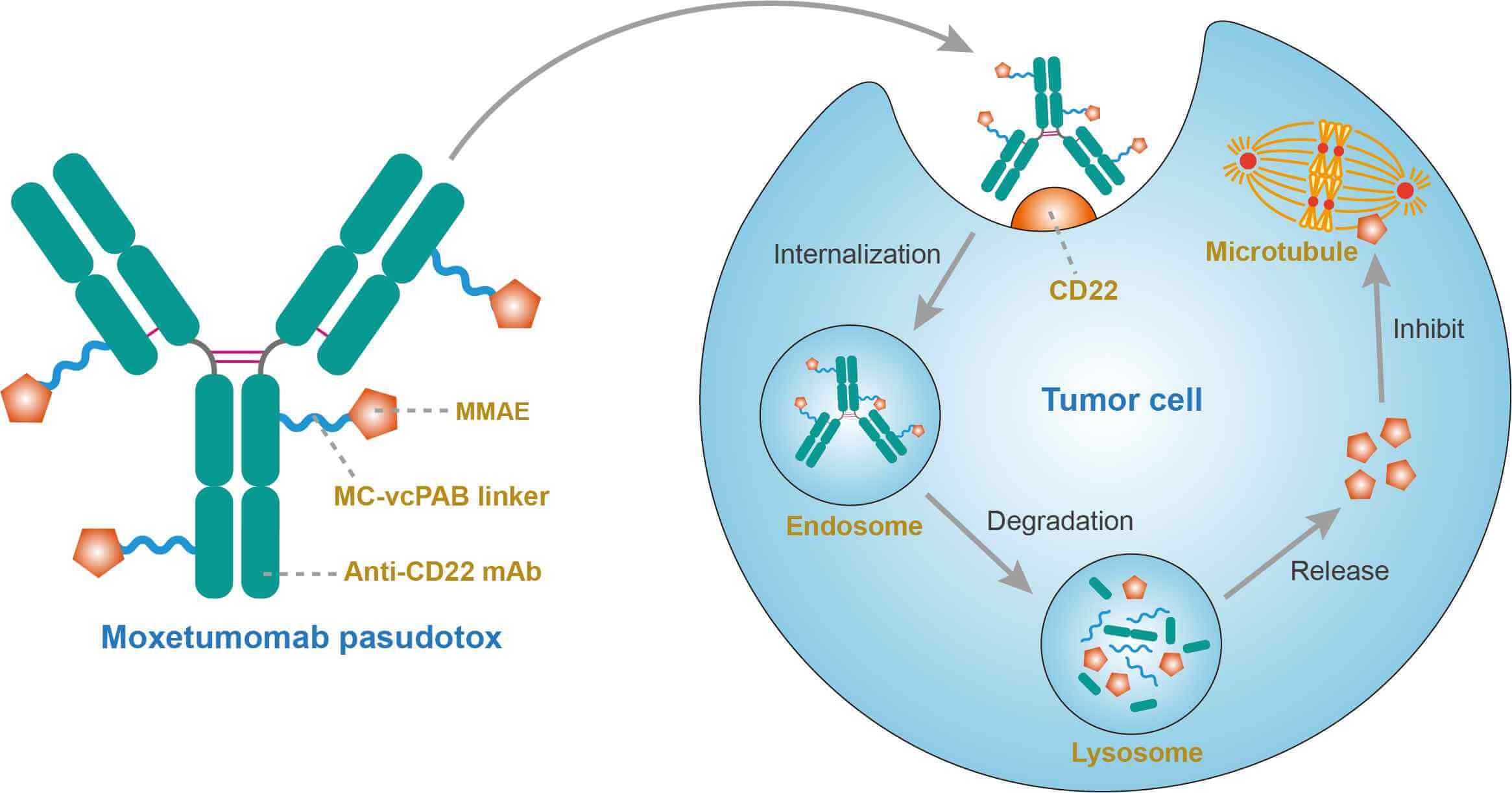
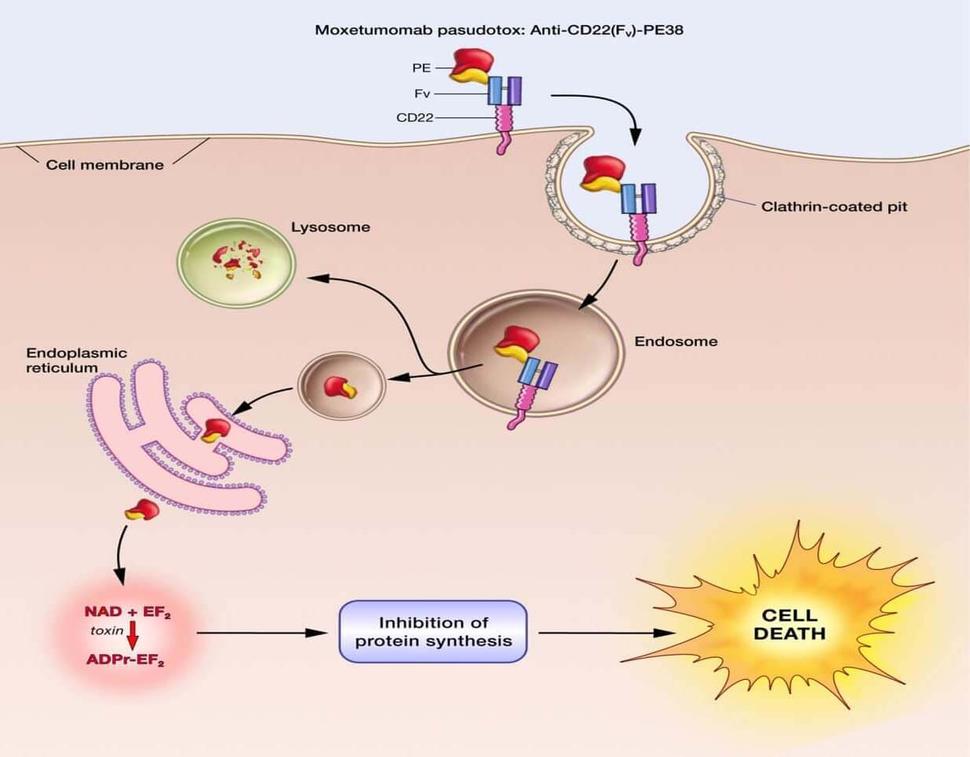
![PDF] Antibody Fusion Proteins: Anti-CD22 Recombinant Immunotoxin Moxetumomab Pasudotox | Semantic Scholar PDF] Antibody Fusion Proteins: Anti-CD22 Recombinant Immunotoxin Moxetumomab Pasudotox | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0e5848fffd0e51622857273f85eab1e135d60d2c/5-Figure3-1.png)
PDF] Antibody Fusion Proteins: Anti-CD22 Recombinant Immunotoxin Moxetumomab Pasudotox | Semantic Scholar
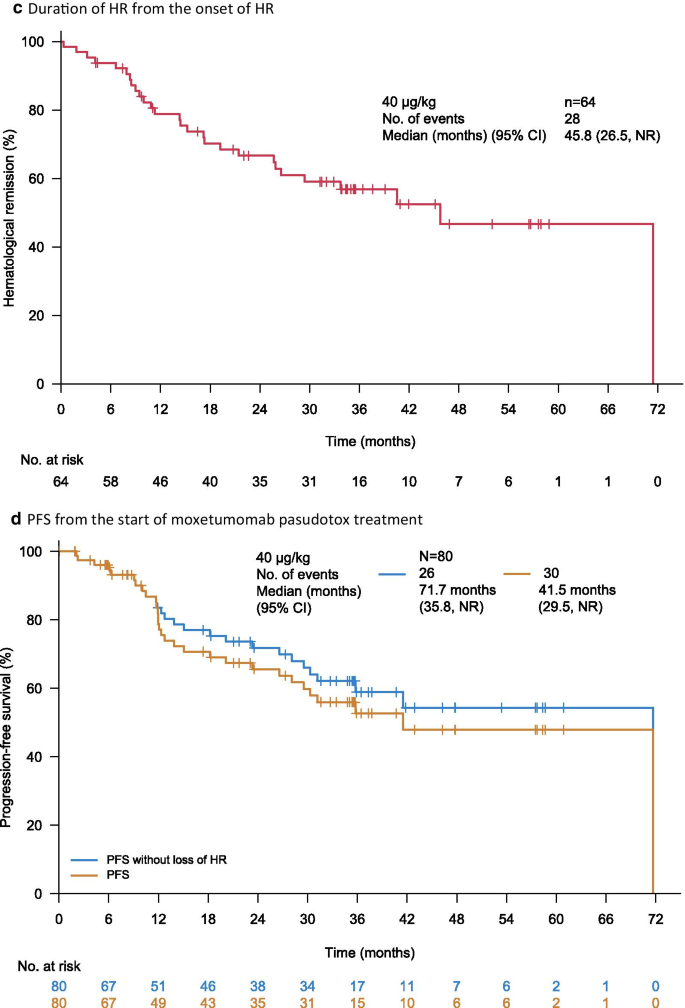
Moxetumomab pasudotox in heavily pre-treated patients with relapsed/refractory hairy cell leukemia (HCL): long-term follow-up from the pivotal trial | Journal of Hematology & Oncology | Full Text

Antibody fusion proteins: anti-CD22 recombinant immunotoxin moxetumomab pasudotox. - Abstract - Europe PMC

Moxetumomab in R/R HCL - Capsule Summary Slidesets - Hematologic Malignancies - 2018 ASCO Annual Meeting - Oncology - Clinical Care Options

Moxetumomab pasudotox provided durable responses in heavily pre-treated... | Download Scientific Diagram

Toxicity of moxetumomab pasudotox. Incidence of toxicity by cycle is... | Download Scientific Diagram
![PDF] Antibody Fusion Proteins: Anti-CD22 Recombinant Immunotoxin Moxetumomab Pasudotox | Semantic Scholar PDF] Antibody Fusion Proteins: Anti-CD22 Recombinant Immunotoxin Moxetumomab Pasudotox | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0e5848fffd0e51622857273f85eab1e135d60d2c/2-Figure1-1.png)
PDF] Antibody Fusion Proteins: Anti-CD22 Recombinant Immunotoxin Moxetumomab Pasudotox | Semantic Scholar
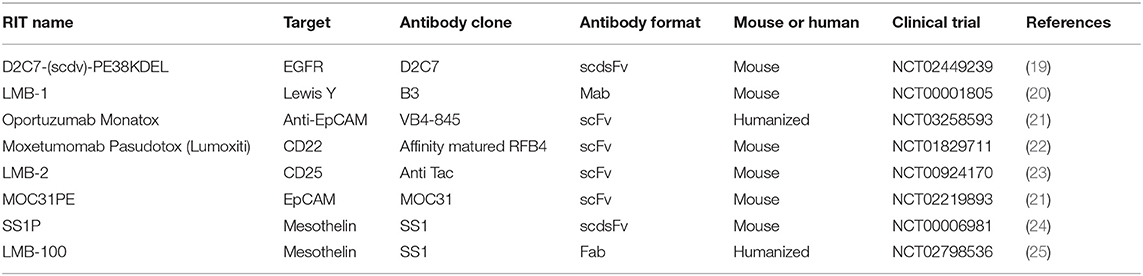

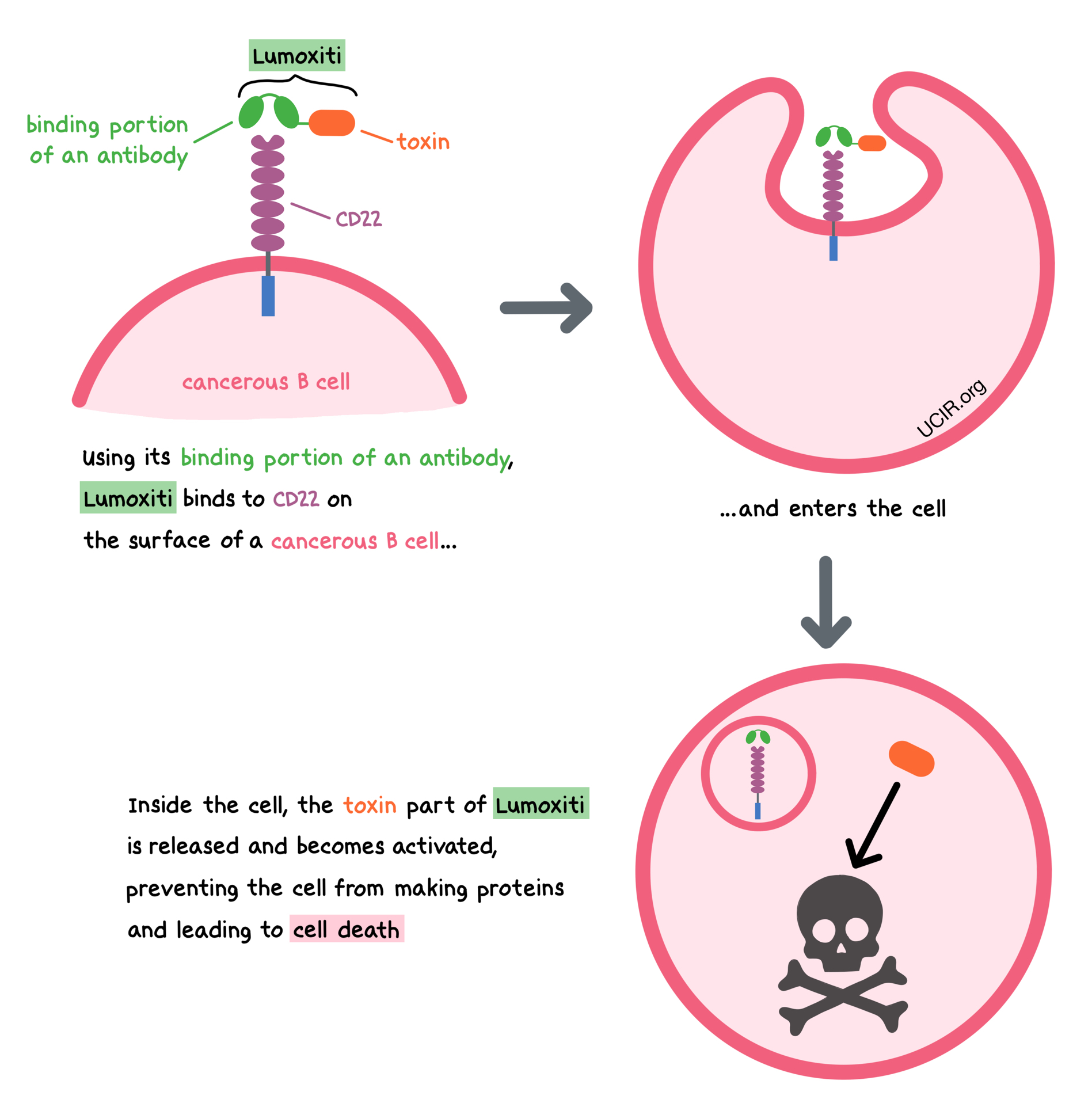





:%0A%0AMoxetumomab%20Pasudotox-tdfk%20for%20Leukemia,%20Hairy%20Cell.png?md=1)